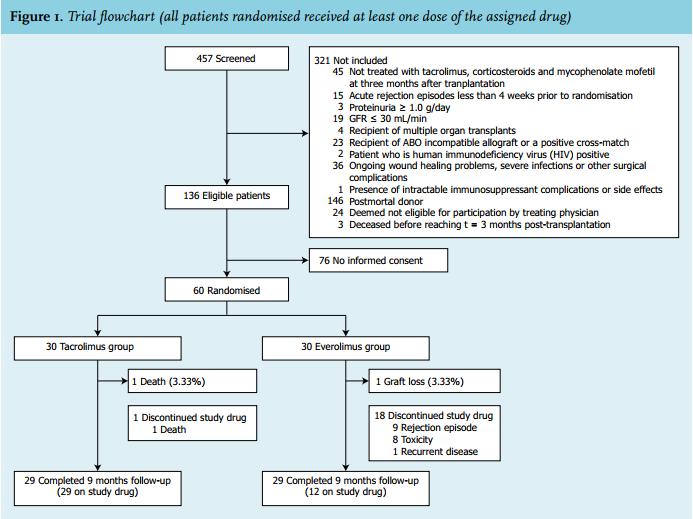
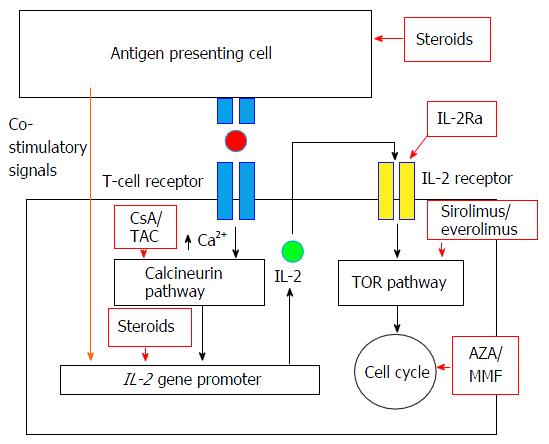
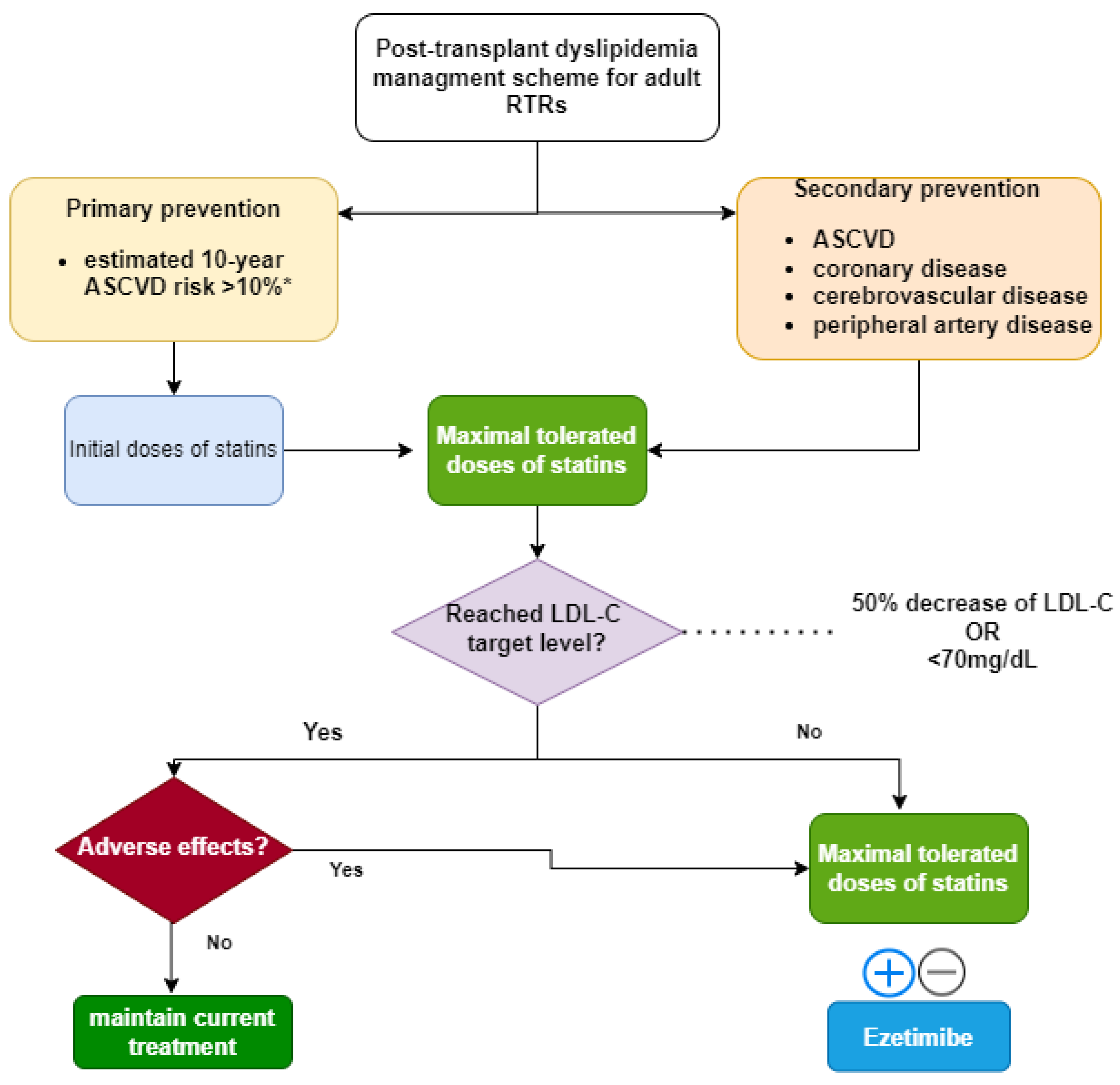
Recommendations for the use of everolimus in de novo kidney transplantation: False beliefs, myths and realities - ScienceDirect
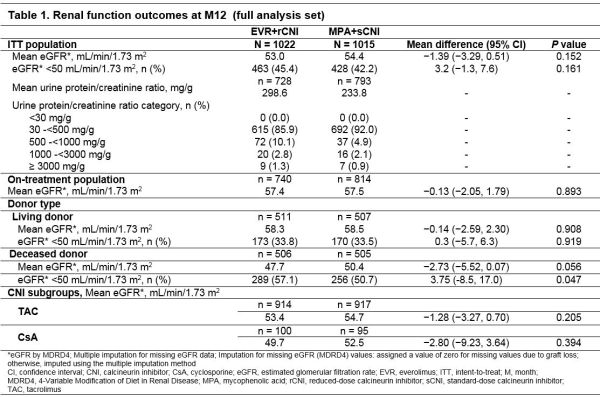
Everolimus with Reduced-Dose Calcineurin Inhibitor versus Mycophenolate with Standard-Dose Calcineurin Inhibitor in De Novo Kidney Transplant Recipients: Renal Function Results from the TRANSFORM Study - ATC Abstracts

Pharmacokinetic principles of dose adjustment of mTOR inhibitors in solid organ transplanted patients - Hartinger - 2022 - Journal of Clinical Pharmacy and Therapeutics - Wiley Online Library

Recommendations for the use of everolimus in de novo kidney transplantation: False beliefs, myths and realities | Nefrología

Recommendations of everolimus use in liver transplant | Gastroenterología y Hepatología (English Edition)
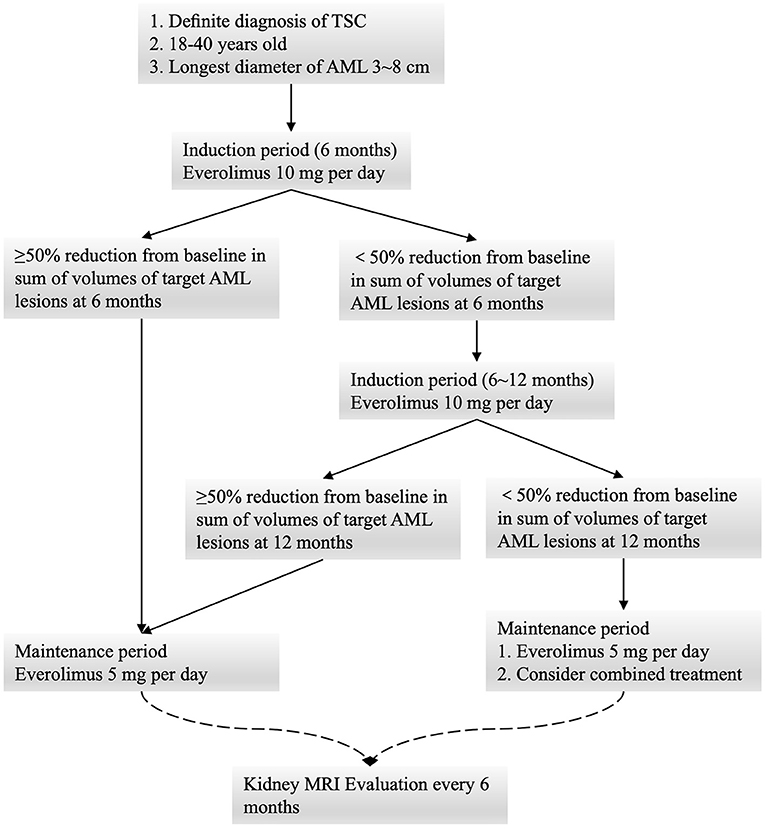
Frontiers | Low-Dose Everolimus Maintenance Therapy for Renal Angiomyolipoma Associated With Tuberous Sclerosis Complex
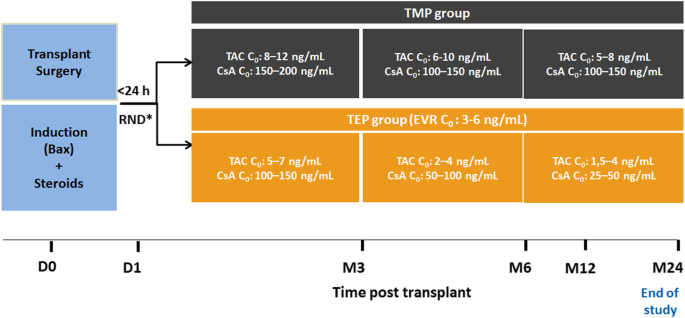
Rationale and design of the OPTIMIZE trial: OPen label multicenter randomized trial comparing standard IMmunosuppression with tacrolimus and mycophenolate mofetil with a low exposure tacrolimus regimen In combination with everolimus in de

Recommendations for the use of everolimus in de novo kidney transplantation: False beliefs, myths and realities | Nefrología

Everolimus-based, calcineurin-inhibitor-free regimen in recipients of de-novo kidney transplants: an open-label, randomised, controlled trial - The Lancet

An overview of the efficacy and safety of everolimus in adult solid organ transplant recipients - ScienceDirect

Everolimus (Certican) in renal transplantation: a review of clinical trial data, current usage, and future directions - ScienceDirect
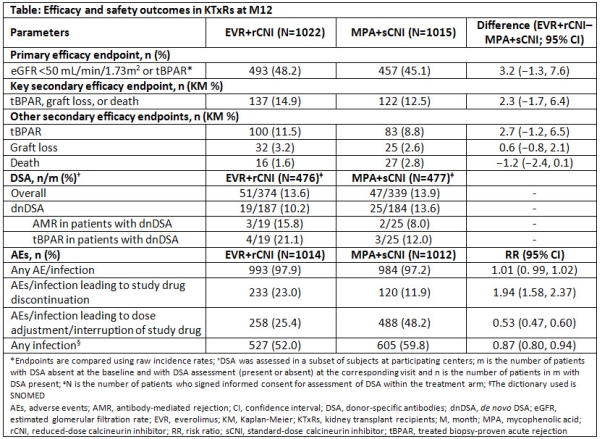
Everolimus with Reduced Calcineurin Inhibitor Exposure in De Novo Kidney Transplant Recipients: Efficacy and Safety Outcomes from the TRANSFORM Study - ATC Abstracts

Early conversion of pediatric kidney transplant patients to everolimus with reduced tacrolimus and steroid elimination: Results of a randomized trial - American Journal of Transplantation
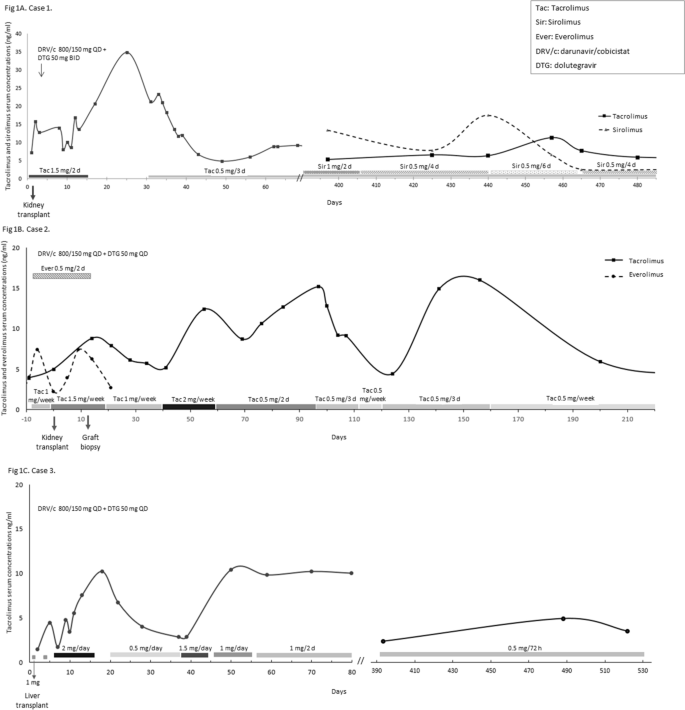
Tacrolimus, Sirolimus and Everolimus Doses in HIV-Infected Solid-Organ Recipients, Requiring a Cobicistat-Based Antiretroviral Regimen: Report of Three Cases and Review | SpringerLink

IJMS | Free Full-Text | Prediction of Tacrolimus Exposure by CYP3A5 Genotype and Exposure of Co-Administered Everolimus in Japanese Renal Transplant Recipients
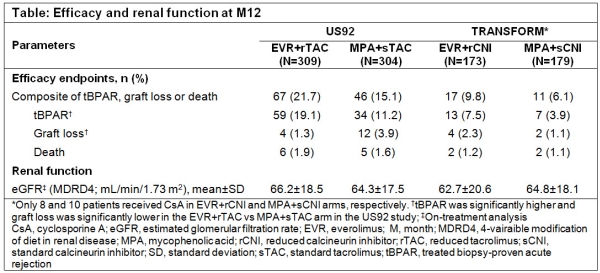
Effect of Early Attainment of Everolimus Target Trough Levels on Efficacy-Safety Outcomes in De Novo Kidney Transplant Recipients: 12-Month Results from US92 and TRANSFORM Studies - ATC Abstracts
Efficacy and Safety of an Everolimus- vs. a Mycophenolate Mofetil-Based Regimen in Pediatric Renal Transplant Recipients | PLOS ONE